 Electronic materials
Electronic materials
Material Sciences at Very High Pressure: Frontier of Mbar Chemistry, a Grant-in-Aid for Specially Promoted Research
Structural Study of 200 K-Superconductivity in Compressed Hydrogen Sulfide In Search of a Material Having Zero Electrical Resistance at Room Temperature- Observation

-
-

-

- Clarifying the crystal structure of 200 K-superconducting hydrogen sulfide
- Conducting measurements under the highly restrictive conditions of ultrahigh pressure
- One step closer toward achieving a room-temperature superconductor that could be the key to solving our energy problems
Discovery of a 200 K superconductor
“Superconductivity” is a phenomenon that the electrical resistance of a material drops to zero as the material is cooled. If used in power transmission lines, superconductive materials can convey electricity with no loss, thereby helping to resolve our energy problems. However, to realize superconductivity, the material must be cooled to an extremely low temperature. As a result, physicists have long been attracted to the idea of discovering and producing a room-temperature superconductor.
It was recently reported that hydrogen sulfide (H2S) demonstrated superconductivity at the relative high temperature of 200 K (approximately -70°C) when compressed to 150 GPa. In order to elucidate the mechanism of the high-temperature superconductivity that could lead to the discovery of materials exhibiting an even higher superconducting transition temperature, information on the crystal structures of sulfur hydride systems under high pressure will be essential. Experiments to replicate superconductivity and research on crystal structures are now being conducted throughout the world, but this research is advancing at a slow pace due to such difficulties of experimentation as generating ultrahigh pressure.
Clarifying the crystal structure of hydrogen sulfide in its superconducting phase
By simultaneously performing electrical resistance measurements to detect the superconductivity and X-ray diffraction of the crystal structure using the high-pressure beamline BL10XU at the large-scale synchrotron radiation facility SPring-8 (Figure 1), we clarified the crystal structure in its superconducting phase. H2S and deuterium sulfide (D2S) were used in the experiments. The samples were compressed to 150 GPa in a diamond anvil cell (DAC) and cooled to 10 K. The resistivity was monitored throughout the cooling process and the critical temperature (zero resistivity) was observed at about 180 K. The X-ray diffraction data collected simultaneously indicated a bcc structure in which the sulfur atom positions suggest formation of H3S (Figure 2). This structure agrees favorably with the theoretically predicted structures in the R3m and Im-3m phases. We concluded that the superconducting phase at 200 K corresponds to the Im-3m phase based on the pressure dependence of the simultaneously measured resistivity. Further, no difference was observed in the crystal structures obtained at the low temperature (10K) and at room temperature. Thus we succeeded in the world’s first clarification that H2S molecules undergo structural change to H3S under high pressure, and that this H3S structure exhibits superconductivity. Further, by simultaneously measuring changes in the transition temperature by pressure, we found that H3S displayed two superconducting phases, as was predicted from theoretical calculations, one with a hexagonal structure and the other with a cubic structure.
Superconductivity at room temperature is theoretically predicted in solid metallic hydrogen under very high pressures exceeding 500 GPa, but hydrogen-derived superconductivity can be anticipated in hydrogen-rich systems under high pressure, such as hydrogen storage alloys and hydrocarbons. These results suggest the potential for superconductivity at even higher temperatures in other hydrogen-rich systems.

Figure 1 A high-pressure device (DAC) housing the sample was inserted into the refrigerator and an XRD experiment combined with electrical resistance measurements was performed as the temperature dropped.
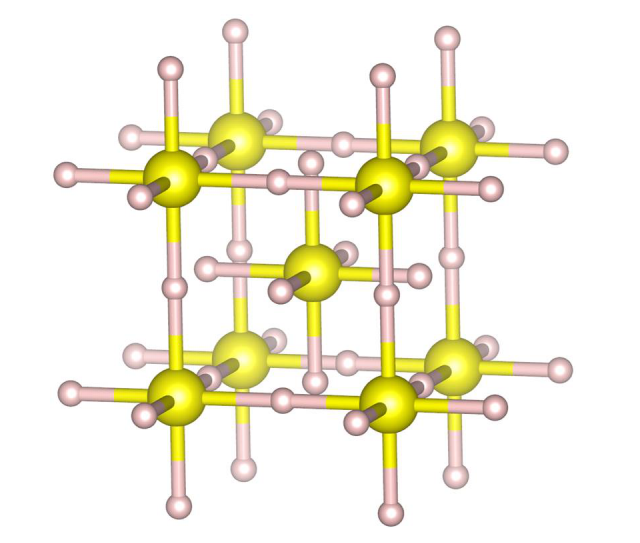
Figure 2 The crystal structure of hydrogen sulfide at the high-temperature superconducting phase (H3S with a cubic structure)

Katsuya Shimizu
Center for Science and Technology under Extreme Conditions
Graduate School of Engineering Science
Osaka University
Research collaborators:
M. Einaga, H. Nakao, and M. Sakata of Center for Science and Technology under Extreme Conditions, Graduate School of Engineering Science, Osaka University; and Y. Oishi and H. Naohisa of the Japan Synchrotron Radiation Research Institute
References:
- [1] A. Drozdov et al., Nature 525, 73 (2015).
- [2] M. Einaga et al., Nature Physics, 12, 835 (2016).
- [3] Duan et al., Sci. Reports 4, 6968 (2014).